A research team recently published a new study on initiating high voltage multi-electron reactions in NASICON cathodes in the journal Energy Material Advances.
“Aqueous secondary batteries hold substantial promise for stationary energy storage,” notes lead researcher Professor Ying Bai, affiliated with the School of Materials Science and Engineering at Beijing Institute of Technology.
“While commercial lithium-ion batteries have witnessed widespread adoption in transportation and various flexible devices over the last three decades, their potential as energy storage sources is on the rise. Nevertheless, the limited availability of lithium and concerns regarding the safety of organic electrolytes underscore the necessity of an ongoing quest for alternatives better suited to stationary energy storage.”
Bai explained that aqueous secondary batteries have attracted attention as the prospective and viable candidates for grid-level energy storage because of the safe aqueous electrolytes in recent years.
“Aqueous zinc-based batteries have garnered significant attention from researchers due to the favorable redox potential and the excellent specific capacity of zinc metal. However, achieving satisfactory performance in practical applications still necessitates further endeavors,” stated Bai.
“Among the various components of the battery, the cathode material plays a pivotal role in determining the electrochemical characteristics of aqueous zinc-based batteries. To date, several categories of electrodes have been employed as cathodes for aqueous zinc-based batteries, such as Prussian blue analogs and manganese-based oxides.”
“Nevertheless, the limited specific capacity of PBAs constrains their broader application in aqueous zinc-based batteries. Furthermore, manganese oxides and vanadium oxides have found widespread use in aqueous zinc-based batteries, initially delivering high capacities ranging from 300 mAh g–1 to 500 mAh g–1. However, due to cathode disproportionation reactions and host structure phase transformations, the cycling lifespan remains inadequate for the practical deployment of aqueous zinc-based batteries.”
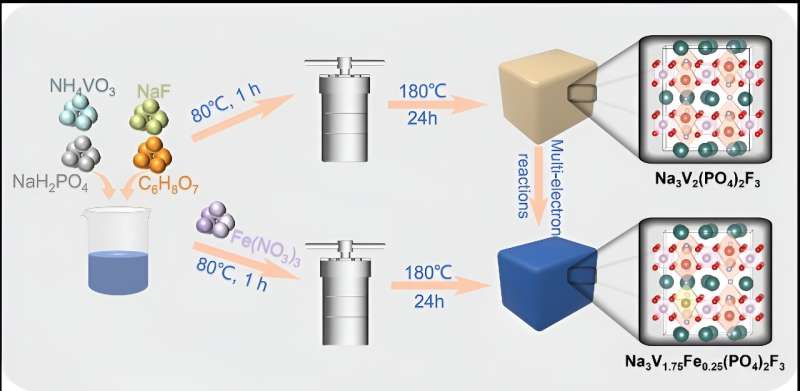
According to Bai, sodium superionic conductors (NASICONs) are considered as competitive electrode materials for aqueous secondary batteries, since they possess a robust polyanionic framework with a general formula of AxM1M2(XO4)3 (A = Li, Na; M1 and M2 = V, Fe, Mn, Ti, Cr; X = P, S).
“The three-dimensional open framework structure not only builds a diffusion pathway for metal ions, but also limits the lattice volume change in the intercalation reaction, which results in good rate performance and cycle lifespan,” Bai said. “The covalent bonding derived from the polyanionic framework and strong induction effects theoretically allow for higher operating voltage, which contributes to the energy density of the NASICONs cathode.”
“Despite the tremendous progress that has been achieved, conventional NASICONs materials in aqueous electrolytes typically have low operating voltages, low energy densities, and poor lifespan, making their applications difficult.”
According to Bai, a typical NASICONs compound, Na3V2(PO4)2F3 (NVPF), has been prepared as the cathode for Zn2+ storage, but its further application is plagued by the structural deterioration and low practical capacity because of the strong electrostatic of Zn2+ with the host material.
“The utilization of multi-ion electrolytes seems to be one of the most effective strategies, in which the cathode material is stabilized by the synergistic interaction of different cations,” Bai said.
“Indeed, NVPF could be utilized as cathode in the aqueous zinc/sodium batteries with a zinc metal anode, where Na+ or Zn2+ is inserted/extracted into/from the NVPF cathode, and Zn2+ is deposited/dissolved onto/from the anode. However, the transition metal dissolution during Na+ or Zn2+ intercalation/extraction still occurs in the aqueous zinc/sodium batteries, inducing the inevitable capacity decay.”
“Transition metal ion substitution represents a promising strategy to enhance structural stability and augment the high potential capacity,” emphasized Bai. “Given the pertinent cost considerations and potential economic advantages, our research team asserts that harnessing low-cost, abundantly available transition metal substitutes is imperative for the advancement of aqueous zinc/sodium batteries featuring NASICONs cathodes.”
More information:
Jiasheng Yue et al, Initiating High-Voltage Multielectron Reactions in NASICON Cathodes for Aqueous Zinc/Sodium Batteries, Energy Material Advances (2023). DOI: 10.34133/energymatadv.0050
Provided by
Beijing Institute of Technology Press Co., Ltd
Citation:
Initiating high voltage multi-electron reactions in NASICON cathodes for aqueous zinc/sodium batteries (2023, September 18)
retrieved 18 September 2023
from https://techxplore.com/news/2023-09-high-voltage-multi-electron-reactions-nasicon.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.
Stay connected with us on social media platform for instant update click here to join our Twitter, & Facebook
We are now on Telegram. Click here to join our channel (@TechiUpdate) and stay updated with the latest Technology headlines.
For all the latest Technology News Click Here
For the latest news and updates, follow us on Google News.
